INTEXT QUESTIONS
Q1. You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution, respectively. If you are given only red litmus paper, how will you identify the contents of each test tube?
Ans. If the colour of red litmus paper gets changed to blue, then it is a base and if there is no colour change, then it is either acidic or neutral. Thus, basic solution can be easily identified.
Let us mark the three test tubes as A, B, and C. A drop of the solution in A is put on the red litmus paper. Same is repeated with solution Band C. If either of them changes colour to blue, then it is basic. Therefore, out of three, one is eliminated. Out of the remaining two, any one can be acidic or neutral. Now a drop of basic solution is mixed with a drop of each of the remaining two solutions separately and then the nature of the drops of the mixtures is checked. If the colour of red litmus turns blue, then the second solution is neutral and if there is no change in colour, then the second solution is acidic. This is because acidic and basic solutions neutralize each other. Hence, we can distinguish between the three types of solutions.
INTEXT QUESTIONS
Q1. Why should curd and sour substances not be kept in brass and copper vessels?
Ans. Curd and other sour substances contain acids. Therefore, when they are kept in brass and copper vessels, the metal reacts with the acid to liberate hydrogen gas and harmful products, thereby spoiling the food.
Q2. Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
Ans. Hydrogen gas is usually liberated when an acid reacts with a metal.
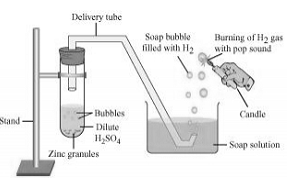
Take few pieces of zinc granules and add 5 ml of dilute . Shake it and pass the gas produced into a soap solution. The bubbles of the soap solution are formed. These soap bubbles contain hydrogen gas.
We can test the evolved hydrogen gas by its burning with a pop sound when a candle is brought near the soap bubbles.
Q3. Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Ans.
INTEXT QUESTIONS
Q1. Why do HCl, , etc., show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Ans. The dissociation of to form hydrogen ions always occurs in the presence of water. Hydrogen ions combine with H2O to form hydronium ions .
The reaction is as follows:
Although aqueous solutions of glucose and alcohol contain hydrogen, these cannot dissociate in water to form hydrogen ions. Hence, they do not show acidic character.
Q2. Why does an aqueous solution of an acid conduct electricity?
Ans. Acids dissociate in aqueous solutions to form ions. These ions are responsible for conduction of electricity.
Q3. Why does dry HCl gas not change the colour of the dry litmus paper?
Ans. Colour of the litmus paper is changed by the hydrogen ions. Dry HCl gas does not contain H+ ions. It is only in the aqueous solution that an acid dissociates to give ions. Since in this case, neither HCl is in the aqueous form nor the litmus paper is wet, therefore, the colour of the litmus paper does not change.
Q4. While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Ans. Since the process of dissolving an acid in water is exothermic, it is always recommended that acid should be added to water. If it is done the other way, then it is possible that because of the large amount of heat generated, the mixture splashes out and causes burns.
Q5. How is the concentration of hydronium ions affected when a solution of an acid is diluted?
Ans. When an acid is diluted, the concentration of hydronium ions per unit volume decreases. This means that the strength of the acid decreases.
Q6. How is the concentration of hydroxide ions affected when excess base is dissolved in a solution of sodium hydroxide?
Ans. The concentration of hydroxide ions would increase when excess base is dissolved in a solution of sodium hydroxide.
INTEXT QUESTIONS
Q1. You have two solutions, A and B. The pH of solution A is 6 and pH of solution B is 8. Which solution has more hydrogen ion concentration? Which of this is acidic and which one is basic?
Ans. A pH value of less than 7 indicates an acidic solution, while greater than 7 indicates a basic solution. Therefore, the solution with pH = 6 is acidic and has more hydrogen ion concentration than the solution of pH = 8 which is basic.
Q2. What effect does the concentration of ions have on the nature of the solution?
Ans. Concentration of can have a varied effect on the nature of the solution. With an increase in ion concentration, the solution becomes more acidic, while a decrease of ion causes an increase in the basicity of the solution.
Q3. Do basic solutions also have ions? If yes, then why are these basic?
Ans. Yes, basic solution also has ions. However, their concentration is less as compared to the concentration of OH” ions that makes the solution basic.
Q4. Under what soil condition do you think a farmer would treat the soil of his fields with quick lime (calcium oxide) or slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
Ans. If the soil is acidic and improper for cultivation, then to increase the basicity of soil, the farmer would treat the soil with quick lime or slaked lime or chalk.
INTEXT QUESTIONS
Q1. What is the common name of the compound ?
Ans. The common name of the compound is bleaching powder.
Q2. Name the substance which on treatment with chlorine yields bleaching powder?
Ans. Calcium hydroxide , on treatment with chlorine, yields bleaching powder.
Q3. Name the sodium compound which is used for softening hard water.
Ans. Washing soda is used for softening hard water.
Q4. What will happen if a solution of sodium hydrocarbonate is heated? Give the equation of the reaction involved.
Ans. When a solution of sodium hydrocarbonate (sodium hydrogencarbonate) is heated, sodium carbonate and water are formed with the evolution of carbon dioxide gas.
Q5. Write an equation to show the reaction between Plaster of Paris and water.
Ans. The chemical equation for the reaction of Plaster of Paris and water can be represented as
NCERT TEXT BOOK EXERCISES
Q1. A solution turns red litmus blue, its pH is likely to be
(a) 1 (b) 4 (c) 5 (d) 10
Ans. (d) Bases turn red litmus blue and acids turn blue litmus red. Basic solution has a pH value more than 7. Since the solution turns red litmus blue, its pH is likely to be 10.
Q2. A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains
(a) NaCl (b) HCl (c) LiCl (d) KCl
Ans. (b) The solution contains HCl.
Q3. 10 mL of a solution of NaOH is found to be completely neutralized by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be
(a) 4 mL (b) 8mL (c) 12 mL (d) 16 mL
Ans. (d) 16 mL of HCl solution will be required.
Q4. Which one of the following types of medicines is used for treating indigestion?
(a) Antibiotic (b) Analgesic (c) Antacid (d) Antiseptic
Ans. (c) Antacid is used for treating indigestion.
Q5. Write word equations and then balanced equations for the reaction taking place when “
(a) dilute sulphuric acid reacts with zinc granules.
(b) dilute hydrochloric acid reacts with magnesium ribbon.
(c) dilute sulphuric acid reacts with aluminium powder.
(d) dilute hydrochloric acid reacts with iron filings.
Ans.
(a) Sulphuric acid + Zinc Zinc sulphate + Hydrogen
(b) Hydrochloric acid + Magnesium Magnesium chloride + Hydrogen
(c) Sulphuric acid + Aluminium Aluminium sulphate + Hydrogen
(d) Hydrochloric acid + Iron Ferric chloride +Hydrogen
Q6. Compounds such as alcohols and glucose also contain hydrogen but are not categorized as acids. Describe an activity to prove it.
Ans. Two nails are fitted on a cork and are kept it in a 100 mL beaker. The nails are then connected to the two terminals of a 6-volt battery through a bulb and a switch. Some dilute HCl is poured in the beaker and the current is switched on. The same experiment is then performed with glucose solution and alcohol solution.
Observations
It will be observed that the bulb glows in the HCl solution and does not glow in the glucose solution.
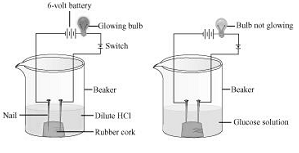
Result
HCl dissociates into and Cl” ions. These ions conduct electricity in the solution resulting in the glowing of the bulb. On the other hand, the glucose solution does not dissociate into ions. Therefore, it does not conduct electricity.
Conclusion
From this activity, it can be concluded that all acids contain hydrogen but not all compounds containing hydrogen are acids.
That is why, though alcohols and glucose contain hydrogen, they are not categorised as acids.
Q7. Why does distilled water not conduct electricity, whereas rain water does?
Ans. Distilled water is a pure form of water and is devoid of any ionic species. Therefore, it does not conduct electricity. Rain water, being an impure form of water, contains many ionic species such as acids and therefore it conducts electricity.
Q8. Why do acids not show acidic behaviour in the absence of water?
Ans. Acids do not show acidic behaviour in the absence of water because the dissociation of hydrogen ions from an acid occurs in the presence of water only. It is the hydrogen ions that are responsible for the acidic behaviour.
Q9. Five solutions A, B, C, D and E when tested with universal indicator showed pH as 4, 1, 11, 7 and 9, respectively. Which solution is
(a) neutral?
(b) strongly alkaline?
(c) strongly acidic?
(d) weakly acidic?
(e) weakly alkaline?
Arrange the pH in increasing order of hydrogen-ion concentration.
Ans.
(a) Neutral Solution D with pH 7
(b) Strongly alkaline Solution C with pH 11
(c) Strongly acidic Solution B with pH 1
(d) Weakly acidic Solution A with pH 4
(e) Weakly alkaline Solution E with pH 9
The pH can be arranged in the increasing order of the concentration of hydrogen ions as: 11 < 9 < 7 < 4 < 1
Q10. Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid is added to test tube B. In which test tube will the fizzing occur more vigorously and why?
Ans. The fizzing will occur strongly in test tube A, in which hydrochloric acid (HCl) is added. This is because HCl is a stronger acid than and therefore produces hydrogen gas at a faster speed due to which fizzing occurs.
Q11. Fresh milk has a pH of 6. How do you think the pH will change as it turns into curd? Explain your answer.
Ans. The pH of milk is 6. As it changes to curd, the pH will reduce because curd is acidic in nature. The acids present in it decrease the pH.
Q12. A milkman adds a very small amount of baking soda to fresh milk.
(a) Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
(b) Why does this milk take a long time to set as curd?
Ans. (a) The milkman shifts the pH of the fresh milk from 6 to slightly alkaline because in alkaline condition, milk does not set as curd easily.
(b) Since this milk is slightly basic than usual milk, acids produced to set the curd are neutralized by the base. Therefore, it takes a longer time for the curd to set.
Q13. Plaster of Paris should be stored in a moisture-proof container. Explain why?
Ans. Plaster of Paris (POP) should be stored in a moisture-proof container because Plaster of Paris, a powdery mass, absorbs water (moisture) to form a hard solid known as gypsum.
Q14. What is a neutralization reaction? Give two examples.
Ans. A reaction in which an acid and base react with each other to give a salt and water is termed as neutralization reaction. In this reaction, energy is evolved in the form of heat.
For example:
(ii) During indigestion (caused due to the production of excess of hydrochloric acid in the stomach), we administer an antacid (generally milk of magnesia, ,
which is basic in nature)
The antacid neutralizes the excess of acids and thus gives relief from indigestion.
Q15. Give two important uses of washing soda and baking soda.
Ans. Two important uses of washing soda and baking soda are as follows:
(1) Washing soda
(a) It is used in glass, soap, and paper industries.
(b) It is used to remove permanent hardness of water.
(2) Baking soda
(a) It is used as baking powder. Baking powder is a mixture of baking soda and a mild acid known as tartaric acid. When it is heated or mixed in water, it releases CO2 that makes bread or cake fluffy.
(b) It is used in soda-acid fire extinguishers.








