Introduction:
Carboxylic acids are organic compounds which contain carboxylic group .
The name carboxyl is derived from two words, i.e. carbonyl and hydroxyl because in carboxylic acid both carbonyl and hydroxyl groups are directly linked to each other.
Carboxylic acids are classified in accordance to the number of –COOH groups present in the molecule.
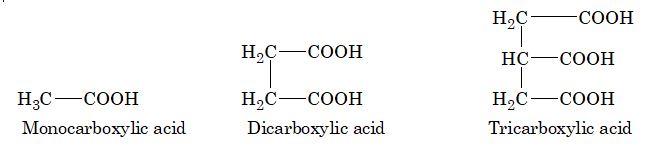
Carboxylic acids are further classified as aliphatic and aromatic. In aromatic, the carboxylic group is attached to aryl group.
Monocarboxylic acid of aliphatic species are commonly known as fatty acid, containing more than twelve carbon atoms, obtained from fats.
GENERAL METHODS OF PREPARATION
From Alcohols
Oxidation of Alcohol: Primary alcohols on oxidation forms monocarboxylic acid with same number of carbon atoms. The oxidation can be carried out in presence of potassium dichromate and dilute sulphuric acid. The initial product of oxidation is the corresponding aldehyde which then forms acid
In case alkaline KMnO4 is used as an oxidizing agent, the aldehyde undergoes oxidation more rapidly than primary alcohol.
The selective oxidation is possible in presence of CrO3 and H2SO4 in acetone. It does not affect CºC and >C=C< but selectively oxidizes hydroxyl group to COOH.
From alkene
(i) Oxidative cleavage: Alkene can be oxidized into corresponding carboxylic acid with hot alkaline KMnO4.
(ii) Carbonylation: (Koch reaction)
When a mixture of alkene, carbon monoxide and steam is heated under pressure at 350°C in presence of phosphoric acid, monocarboxylic acid is formed.
From alkyne:
On oxidation with chromic acid (K2CrO4 + H2SO4) single carboxylic acid is formed
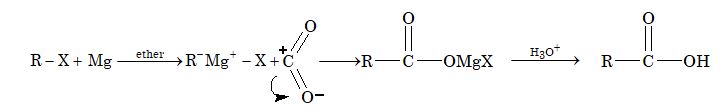
By Carbonation of Grignard Reagents
Grignard reagent on treatment with carbon dioxide forms a addition product, which on hydrolysis gives acid.
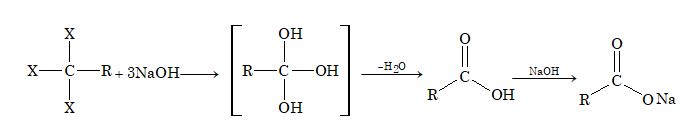
Hydrolysis of Trihalogen Derivatives
Trihalogen derivatives of alkanes in which all the three halogen atoms are attached to same carbon atom, on hydrolysis with aqueous alkali solution, form mono carboxylic acids
Heating Sodium Alkoxide with Carbon Monoxide
Monsanto Process:
Methanol reacts with carbon monoxide in the presence of small amount of rhodium (III) salt, HI and H2O to give acetic acid.
Hydrolysis of Acid Derivatives
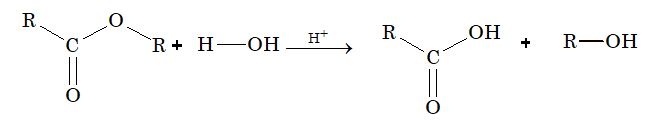
(i) From ester:
(ii) From acid amide:
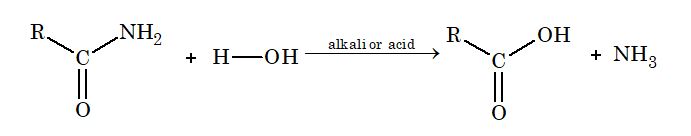
(iii) From anhydrides:
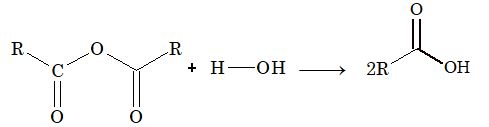
(iv) From acid halide:
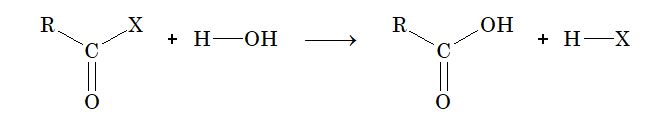
By Hydrolysis of Alkane Nitrile
The compound containing C≡N group can be hydrolysed to carboxylic acid with an additional carbon atom than original alkyl halide.
Illustration 1: End product of the following sequence of reaction is
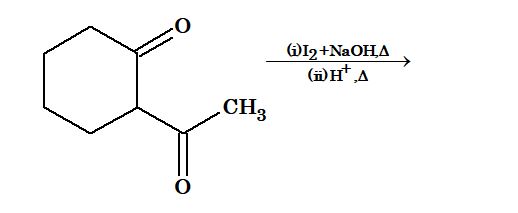
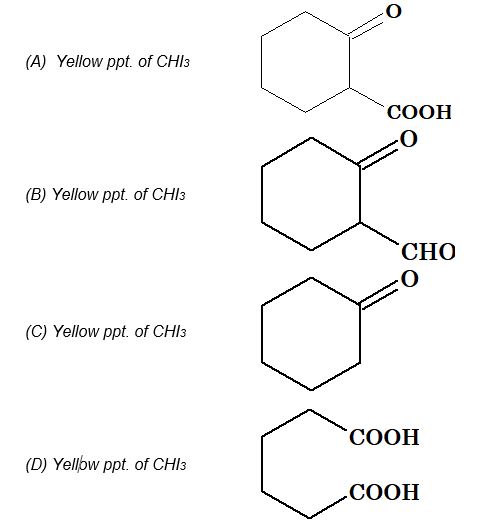
Solution: (C). Intermediate is

PHYSICAL PROPERTIES
1. The lower fatty acids upto C10 are colourless liquids. The higher ones are waxy solids.
2. The molecules of carboxylic acid are polar in nature and exhibit hydrogen bonding.
3. The lower members are highly soluble in water but the solubility decreases with the rise of molecular mass. The acids above C10 carbon are insoluble in water. It is due to its polar nature and tendency to form H – bond.
4. Boiling point of carboxylic acids increase regularly with increase in molecular mass. Boiling points of acid are higher than alcohol and ether. This is due to intermolecular hydrogen bonding between acid molecules. It exists in dimeric form
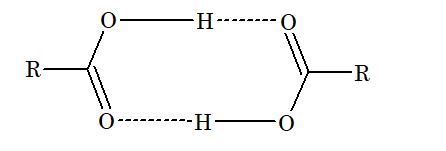
Illustration 2: The correct statement about A, B & C is
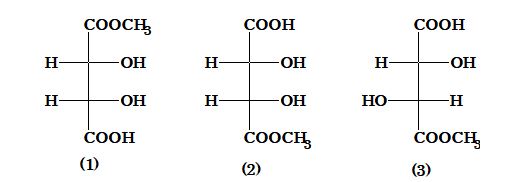
(A) 1 & 2 are identical
(B) 1 & 2 are diastereomers
(C) 1 & 3 are enantiomers
(D) 1 & 2 are enantiomers
Solution: (D). Because (A) & (B) both are non super imposable mirror image so they are enantiomers.
CHEMICAL PROPERTIES
The reactions of carboxylic acid can be studied under four different categories
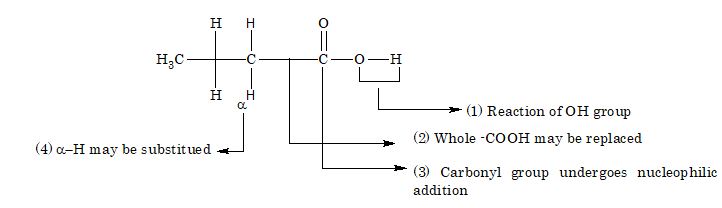
Reaction involving Alkyl Group
α-hydrogen atom undergoes substitution due to presence of electronegative carbonyl group
α – halogenation of acid (Hell Volhard Zelinski Reaction):

Direct iodination can be done in the presence of an oxidizing agent like iodic acid or mercuric oxide.

Oxidation: Monocarboxylic acids except formic acid are resistant to oxidation. But at higher temperature it oxidizes into carbon dioxide and water.
In presence of H2O2 and Ag2O, it is oxidized to β – hydroxy acid.
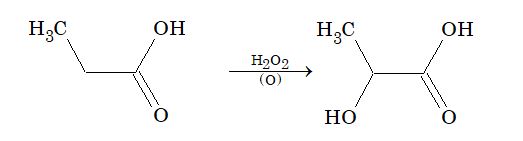
Reaction Involving Carboxyl Group
Salt formation: Carboxylic acids are weak acids and their carboxylate ions are strong conjugated bases. They are slightly alkaline due to hydrolysis of carboxylate ions. The order of acidity and basicity of corresponding conjugated bases are follows:
Acidity: RCOOH > HOH > R-OH > CH≡CH > NH3 > RH
Basicity: R – COO– < OH– < RO– < CH≡C– < NH2– < R–
Carboxylic acids react with metal to liberate hydrogen. It is soluble in both NaOH and NaHCO3 solution.
2CH3 -COOH + 2Na → 2CH3–COO−Na+ + H2
CH3 – COOH + NaOH → CH3–COO−Na+ + H2O
CH3 – COOH + NaHCO3 → CH3 – COO−Na+ H2O + CO2
Acidity of carboxylic acids:
The strength of an acid depends on the ability of acid to lose proton. When dissolved in water it is expressed in terms of dissociation constant (Ka)
Acid strength Ka value
Cause of acidic nature:
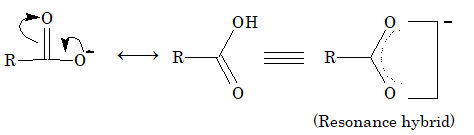
The acidity of carboxylic acid is due to resonance stabilization of its anion.
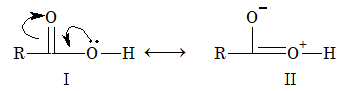
Due to electron deficiency on oxygen atom of the hydroxyl group (structure – II), there is displacement of electron pair of O–H bond towards oxygen atom. This help in release of hydrogen as proton. The resulting carboxylate ion is also stabilized by resonance as resonance energy of the carboxylate ion is much higher than that of undissociated acid.
Effect of substitution on acidity:
1. Acidic strength -I effect (electron withdrawing group)
2. Acidic strength
3. Increase in distance of halogen atoms from COOH group decreases acidity.
4. Increase in electronegativity of halogen atoms increases the acidity.
Effect of electron withdrawing groups (–I effect):
(i) An electron with drawing group stabilizes the anion by dispersing the negative charge and therefore increases acidity.
Cl3C–COOH > Cl2 CH–COOH > Cl-CH2COOH > CH3COOH
(ii)
(iii) F – CH2COOH > BrCH2COOH > ICH2 COOH
Effect of electron releasing group (+I effect):
Electron releasing group increase electron density on oxygen atom resulting decrease in stability and decrease acidic strength.
Illustration 3: Which of the following does not undergo Hell – Volhard Zelinsky reaction?
(A) HCOOH (B) CCl3COOH (C) C6H5COOH (D) All
Solution: (D). None of these contain alpha H atom
Illustration 4: Among the compounds
the correct order of decreasing acidity is
(A) I > II > III > IV (B) IV > III > II > I (C) IV > III > I > II (D) III > IV > I > II
Solution: (C). More the ‘s’ character of the C adjacent to -COOH group, more will be the electronegativity of that carbon and hence more will be the acidity.
Reaction with ketene:
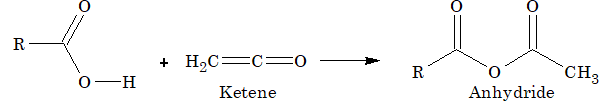
Reaction with diazomethane:
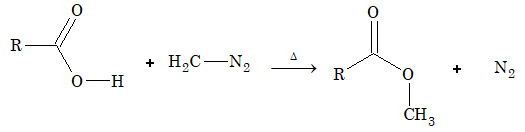
Reactions Involving Replacement of OH
Formation of acid halide:
CH3COOH + PCl5 CH3COCl + HCl + POCl3
3CH3COOH + PCl3 3CH3COCl + H3PO3
Reaction with alcohol (esterification):
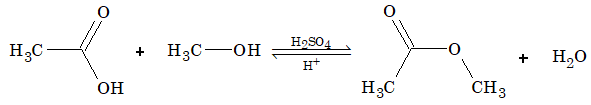
This reaction is reversible and reaches equilibrium when there are enough amount of reactant & product.
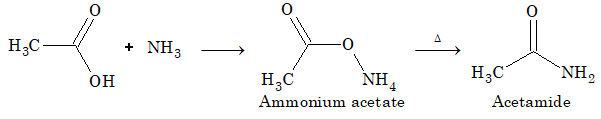
Reaction with ammonia
Reaction with P2O5
Reduction: The COOH group is reduced to CH2OH group in presence of LiAlH4.
With LiAlH4 the – COOH is reduced to – CH2 – OH.
On heating with HI & red P, product is alkane
Catalytic reduction gives alkane
R – COOH + LiAlH4 R – CH2 – OH + H2O
In presence of red phosphorus at 200°C, COOH group is reduced to alkane.
R – COOH + 6HI R – CH3 + 2H2O + 3I2
The product is again alkane when COOH group is catalytically reduced in presence of Ni.
R – COOH + 3H2 R – CH3 + 2H2O
Illustration 5: End product of this conversion is
Solution: (A). NaBH4 reduces reactant to
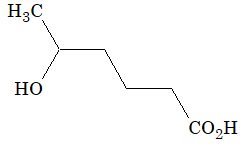
which forms cyclic ester
Illustration 6: Propanoic acid
In above sequence Y is
(A) phenyl ethyl ketone
(B) phenyl acetate
(C) ethyl benzene
(D) ethyl benzoate
Solution: (A).
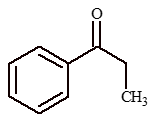
Reaction Involving COOH as a Whole
Decarboxylation: It is a process of elimination of CO2 from a salt of carboxylic acid, in presence of soda lime and the product is alkane.
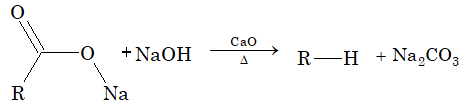
Dibasic acids with two – COOH groups on same carbon atom will loose a molecule of CO2 on simple heating.
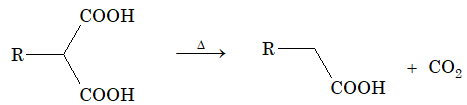
Decarboxylatoin of a-keto acids will take place with warm dil. H2SO4 to form aldehyde.
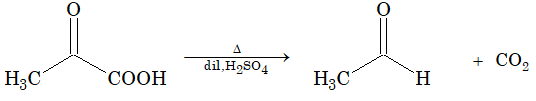
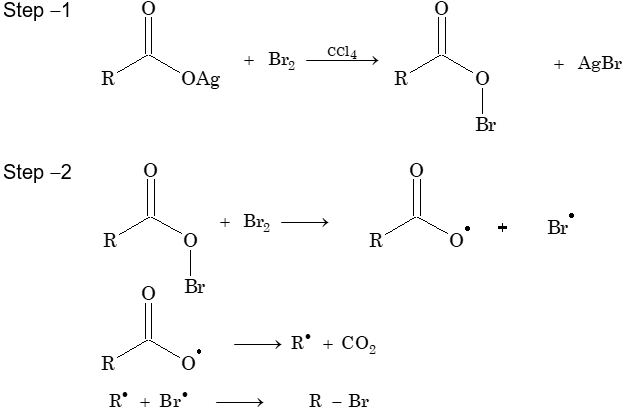
Hunsdiecker reaction: An alky halide is formed when the silver salt of carboxylic is heated with halogen
A carboxyl radical is produced in a two-step process:
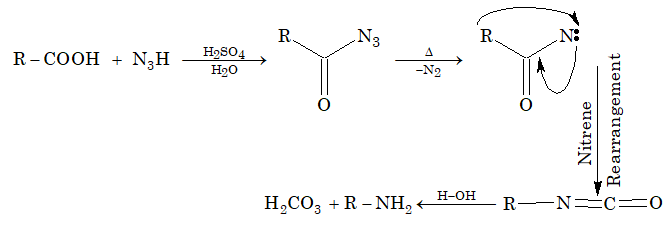
Formation of amine (Schmidt reaction): Primary amine containing one carbon atom less than the acid are formed when monocarboxylic acid react with hydrazoic acid (N3H) in presence of concentration H2SO4.
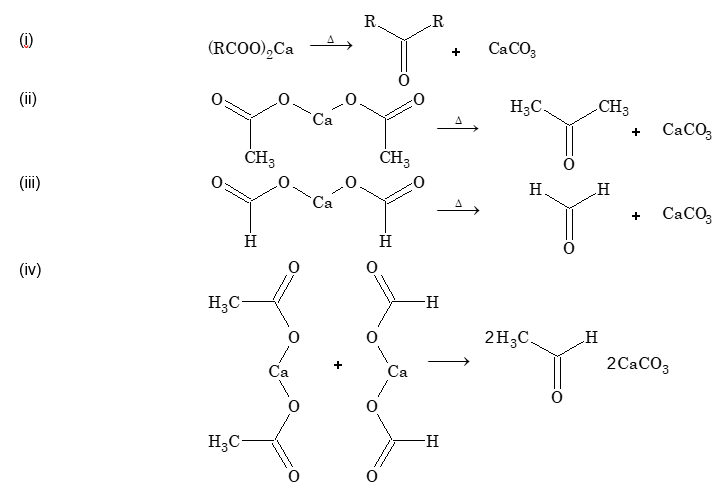
Dry distillation of acid (Heating of calcium salt of acid): When calcium salt of fatty acids other than calcium formate are heated, ketones are formed. When calcium formate is heated, formaldehyde is formed. When a mixture of calcium salt of a fatty acids and calcium formate is heated, aldehyde is formed.
Reaction chart of acetic acid
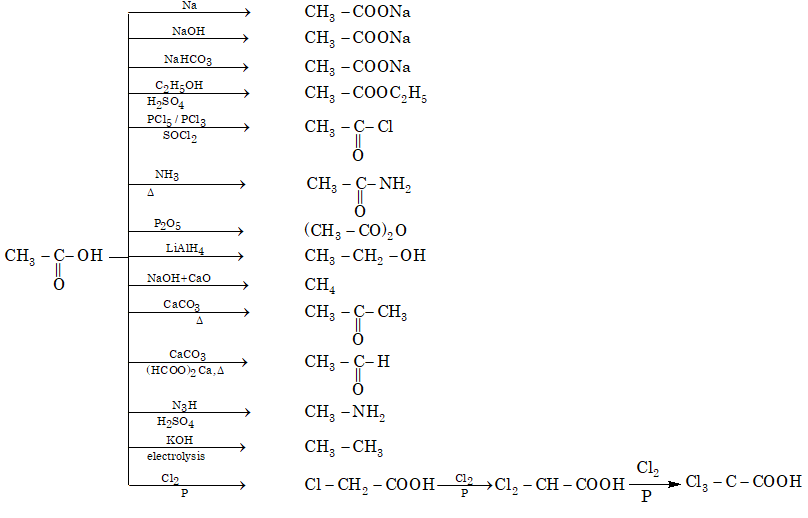
Identification Test
|
Acetic acid Reducing nature |
Formic acid |
|
|
1. Tollens reagent |
Unaffected |
Gives silver mirror or black ppt. HCOOH+Ag2O 2Ag¯ + CO2 + H2O |
|
2. Fehling solution |
Unaffacted |
Gives red ppt. HCOOH + 2CuO Cu2O + CO2 + H3O |
|
3. Mercuric chloride |
Unaffacted |
Form white ppt. which change to Greyish black. HgCl2 Hg2Cl2 2Hg |
|
4. Acidic KMnO4 |
Unaffected |
Decolourize. |
|
5. Acid + NaHSO3 + Sodium nitroprusside |
Unaffected |
Greenish blue colour. |
|
Acid + FeCl3 |
Wine red |
Red colour which changes to brown ppt. on heating |
Illustration 7: Which of the following carboxylic acids is the most reluctant to form ester with a given alcohol in the presence of a catalytic amount of concentrated H2SO4?
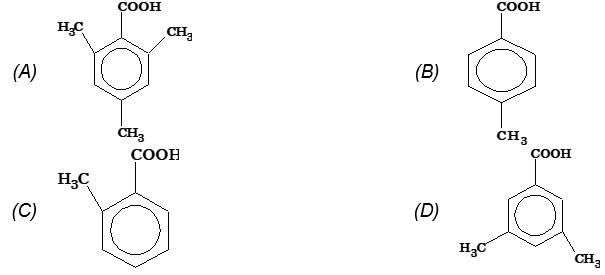
Solution: (A). The nucleophilicity of carboxylic carbon is reduced due to presence of electron releasing -CH3 groups at ortho and para-positions of benzoic acid.
Acid derivatives
The classes of compounds obtained from acids by the replacement of –OH group of –COOH group with – X, – NH2, -OOCR or – OR are termed as acid derivatives
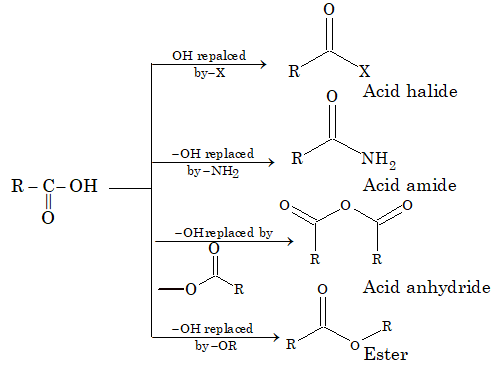
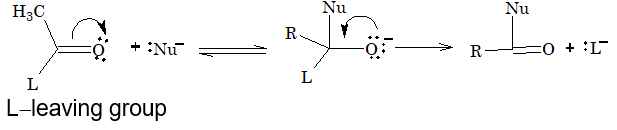
Acyl derivatives are characterized by nucleophilic substitution reactions.
ACETYL CHLORIDE
Method of Preparation
Acetyl chloride is prepared by heating acetic acid with phosphorous trichloride, phosphorus pentachloride or thionyl chloride. Thionyl chloride is better best reagent because the byproducts are in gaseous forms, which are easily separated.
(i)
(ii)
(iii)
Physical Properties
1. It is a colourless liquid with a pungent odour.
2. It is soluble in ether acetone and acetic acid.
Chemical Properties
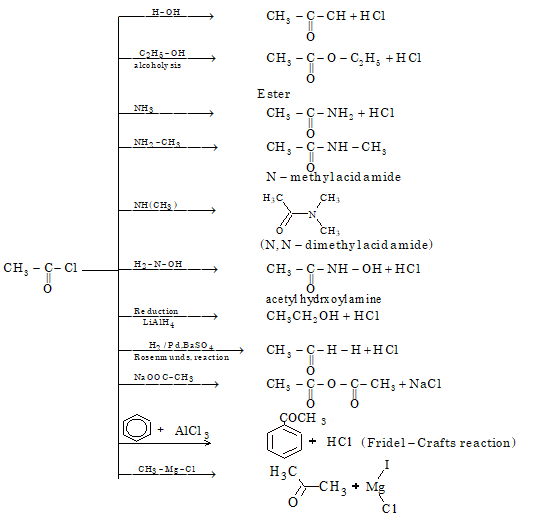
ACETIC ANHYDRIDE
Methods of Preparation
1. It is prepared by heating glacial acetic acid with a dehydrating agent like P2O5 or anhydrous ZnCl2.
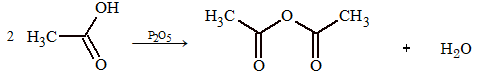
2. Acetic anhydride is prepared by heating sodium salt of acid with acid halide.
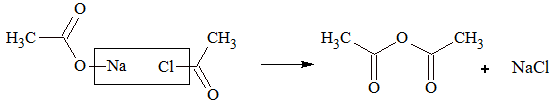
Physical Properties
1. It is colourless, pungent smelling liquid. It boils at 139.5° C.
2. It is sparingly soluble in water but soluble in ether, alcohol and acetic acid.
Chemical Properties
The reactivity of anhydride is less than acid halide. One half of the acetic anhydride is used for acetylation while other half is converted into acetic acid.
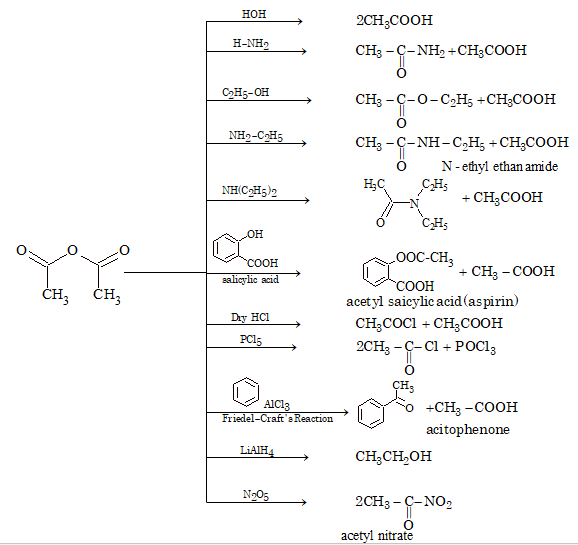
Illustration 8: The formula C4H4O4 can represent
(A) A cyclic ester of dibasic acid
(B) A cis–dibasic acid
(C) A – Trans dibasic acid
(D) All are correct
Solution:(D).
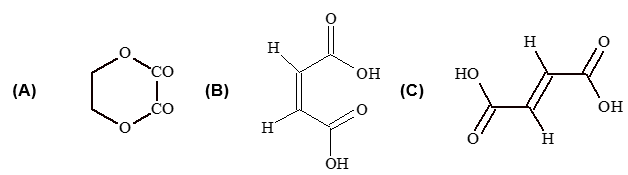
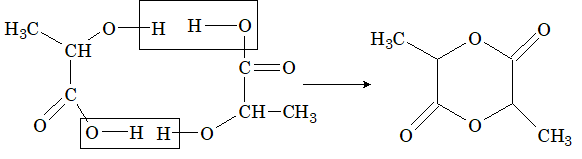
Illustration 9: Lactic acid, CH3CHOHCOOH, when heated strongly gives mainly

Solution: (A)
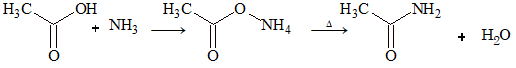
ACETAMIDE
Methods of Preparation
(i) Laboratory preparation: Acetic acid on reaction with ammonia gives ammonium acetate, which on heating gives acetamide.
(ii) By ammonolysis: All acid derivatives on ammonolysis gives acetamide.
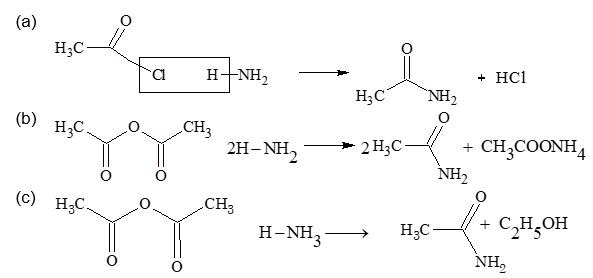
(iii) By partial hydrolysis of cyanide: Alkyl cyanide on hydrolysis in presence of alkaline H2O2 or by dil. H2SO4 gives acetamide.
Physical Properties
1. Acetamide is a colourless crystalline solid, it is readily soluble in water and alcohol.
2. It exist as a dimer due to hydrogen boding
Chemical Properties
(i) Hydrolysis: Acetamide is hydrolysed by water, rapidly by acids and a more rapidly by alkalies.
(ii) Amphoteric nature: Acetamide behaves as an amphoteric compound i.e. it shows acidic as well as basic nature. If forms salt with acids and bases both.
(iii) Reduction: Acetamide on reduction in presence of sodium and ethanol or ethereal solution of LiAlH4 gives alkyl amine.
(iv) Dehydration: When heated with P2O5 or POCl3, it forms alkyl cyanide.
(v) Action of nitrous acid:
(vi) Action with PCl5: When heated with PCl5, acetamide forms methyl cyanide
(vii) Hofmann bromide reaction or Hofmann degradation: Amides when heated with bromine and caustic soda, yield primary amines containing one carbon atom less than amide.
The reaction occurs in tree stages:
1.
2.
3. CH3NCO + 2KOH CH3NH2 + 2KBr + K2CO3 + 2H2O
Illustration 10: Acetic acid is allowed to react with NH3 to form a compound (A). The latter on strong heating produces a compound (B). (B) is
(A) acetonitrile
(B) ammonium acetate
(C) acetamide
(D) aminoacetic acid
Solution: (C).
FORMULAE AND CONCEPTS AT A GLANCE
1. Monocarboxylic acids are called fatty acids.
2. Carboxylic acid are liquids/solids at room temperature. Formic acid exists as dimer in vapour state and as a polymer in liquid and solid state.
3. Even membered carboxylic acids have markedly higher melting points than odd members preceeding or following it.
4. Fatty acids are weaker than inorganic acids due to resonance stabilization in fatty acids.
5. The presence of electron withdrawing substituents, – NO2 – C6H5, – C º N1 – NO2 etc, enhance the acidic character while electron releasing groups are Me, Et, etc, decrease the acidic strength.
6. In esterification of acids, the rate of esterification follows the order
(i) CH3OH > CH3CH2OH > (CH3)2CHOH > (CH3)3COH
(ii) HCOOH > CH3COOH > (CH3)2CHCOOH > (CH3)3CCOOH
7. – COOH group is deactivating and m directing. Deactivation by this group is so strong that aromatic acids do not undergo Friedal craft reaction.
8. Nucleophilic substitution at acyl carbon in acid derivative is faster than in alkyl halides, due to presence of > C = O group.
9. The relative reactivity of acid derivatives towards nucleophilic substitution reaction is .
10. Acid derivatives although contain > C = O group, yet do not undergo the usual properties of carbonyl group due to the presence of resonance.
SOLVED PROBLEMS-1
Prob 1: What is the product when dry ice react with ethyl magnesium bromide followed by hydrolysis?
Sol:
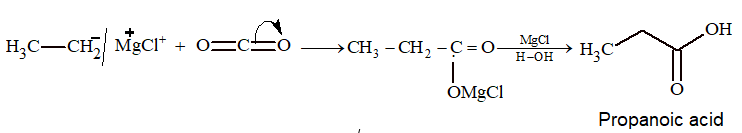
Prob 2: What is Z in the following sequence of reactions?
Sol:
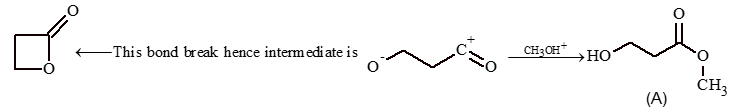
Prob 3: What is A in the following reaction
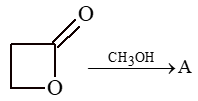
Sol:
Prob 4: R—CH2—CH2OH can be converted in R—CH2CH2COOH. What are the reagents in this conversion.
Sol:
RCH2CH2OH RCH2CH2Br RCH2CH2CN RCH2CH2COOH
Thus, reagents required are PBr3, KCN, H–.
Prob 5: On subjecting mesityl oxide to the iodoform reaction, one of the products is the sodium salt of an organic acid. What is the structure of this acid?
Sol:
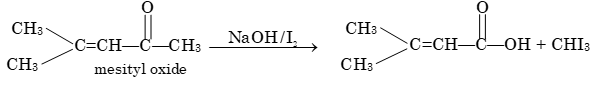
Prob 6: Given three acids
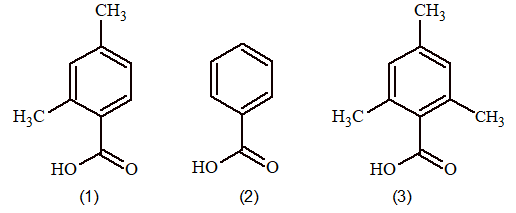
What is the correct order of ease of acid catalysed esterification?
Sol: Catalysed esterification involves nucleophilic attack of alcohol molecule on the protonated acid molecule. The crowding of electron releasing alkyl group of -C atom make the acid less susceptible to nucleophile attack so order is 2>1> 3
Prob 7: What are A and B in the following reactions?
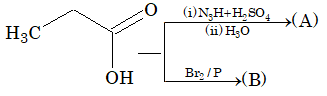
Sol: (i)Formation of (A) the product is obtained by curtius reaction that is amine
CH3 –CH2–NH2
(ii) In formation of (B) a halogenation takes place according to H.V.Z reaction.
Prob 8: What is B in the following reaction
Prob 9: How will you distinguish between formic acid and acetic acid?
Sol: (i) Formic acid produces silver mirror with ammonical silver nitrate solution. Acetic acid does not give this test.
(ii) Formic acid forms a white precipitate with HgCl2 solution. Acetic acid does not respond to this test.
Prob 10: Two compounds possess the same empirical formula CH2O. The vapour density of compound (A) is 15 while that of (B) is 30, (A) reduced Fehling’s solution but did not react with NaHCO3 while (B) did not reduce Fehling’s solution but produced effervescences with NaHCO3. Deduce the structural formulae of (A) and (B)
Sol: Empirical mass = 30
Molecular mass of (A) = 2 15 = 30 Molecular mass of (B) = 30 2 = 60
Molecular formula of (A) = CH2O;Molecular formula of (B) = C2H4O2
From reactions, it is evident that (A) is an aldehyde and (B) is an acid (A) is, therefore, HCHO (formaldehyde) and (B) is CH3COOH (acetic acid).
SOLVED PROBLEMS-2
Prob 1: When acetic acid reacts with ketene, product formed
(A) ethyl acetate
(B) aceto – acetic ester
(C) acetic anhydride
(D) no reaction
Sol: (C).
Prob 2: The pKa of acetylsalicylic acid (aspirin) is 3.5. The pH of gastric juice in human stomach is about 2-3 and pH in the small intestine is about 8. Aspirin will be.
(A) Unionized in the small intestine and in the stomach
(B) Completely ionized in the stomach and almost unionized in the small intestine.
(C) Ionized in the stomach and almost unionized in the small intestine
(D) Ionised in the small intestine and almost unionised in the stomach
Sol: (D). More ionized in basic medium and less ionized in acidic medium, common ion effect
Prob 3: The ease of alkaline hydrolysis is more for

Sol:
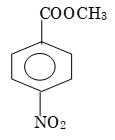
Then is more electron deficiency on carbonyl carbon.
Prob 4: Which of the following acid is capable to produce three series of salts?
(A) Oxalic acid (B) Citric acid (C) Tartaric acid (D) Salicylic acid
Sol: (B). Citric acid is tribasic so it can produce three series of salt.
Prob 5: Acetone & sodium hypochlorite reacts to give
(A) Monochloro acetone
(B) Chloro acetone
(C) chloroforms
(D) chloretone
Sol: (C).
Prob 6: CH3 – COOH + HCOOH Main product of reaction is
(A) CH3 – CHO (B) CH3 – COCH3 (C) HCHO (D) (CH3CO)2O
Sol: (A). OH heating in the presence of MnO at 300°C decarboxylation occurs producing an aldehyde
CH3 – COOH + H – COOH CH3 – CHO + CO2 H3O
Prob 7: CH3 – CH2 – ONa A
(A) CH3 – CH2 – COOH in both
(B) CH3 – CH2– CHO in both
(C) CH3 – CH2 – COOH, CH3 – CHO
(D) CH3 – CHO; CH3 – COOH
Sol: (A) & (B). Heating alkoxide with Co produce acid. In second cast reaction is called oxo process given mixture of acidic.
Prob 8: The amide anode is converted into acid with evolution of nitrogen gas by
(A) HCl (B) HNO2 (C) NaOH (D) P2O5
Sol: (B).
Prob 9: It is difficult to esterify R3C – COOH because of
(A) steric hindrance
(B) delocalization
(C) Inductive effect of R – Group
(B) Hyper conjugation of alkyl group
Sol: (A). Due to steric effect of alkyl group release of H+ ion become difficult so it can’t esterify.
Prob 10: Preparation of alkyl bromide by silver salt of acid collectively known as
(A) de carboxylic + elimination
(B) de carboxylation + halogenation
(C) Dehydration + decarboxylation
(D) None
Sol: (B).
Due to removal by CO2 halogenation of alkyl group reaction is called decarboxylation & halogenation.








